This guide gathers practical dietary and behavioral recommendations useful for reducing the factors that may promote a state of chronic low-grade inflammation.
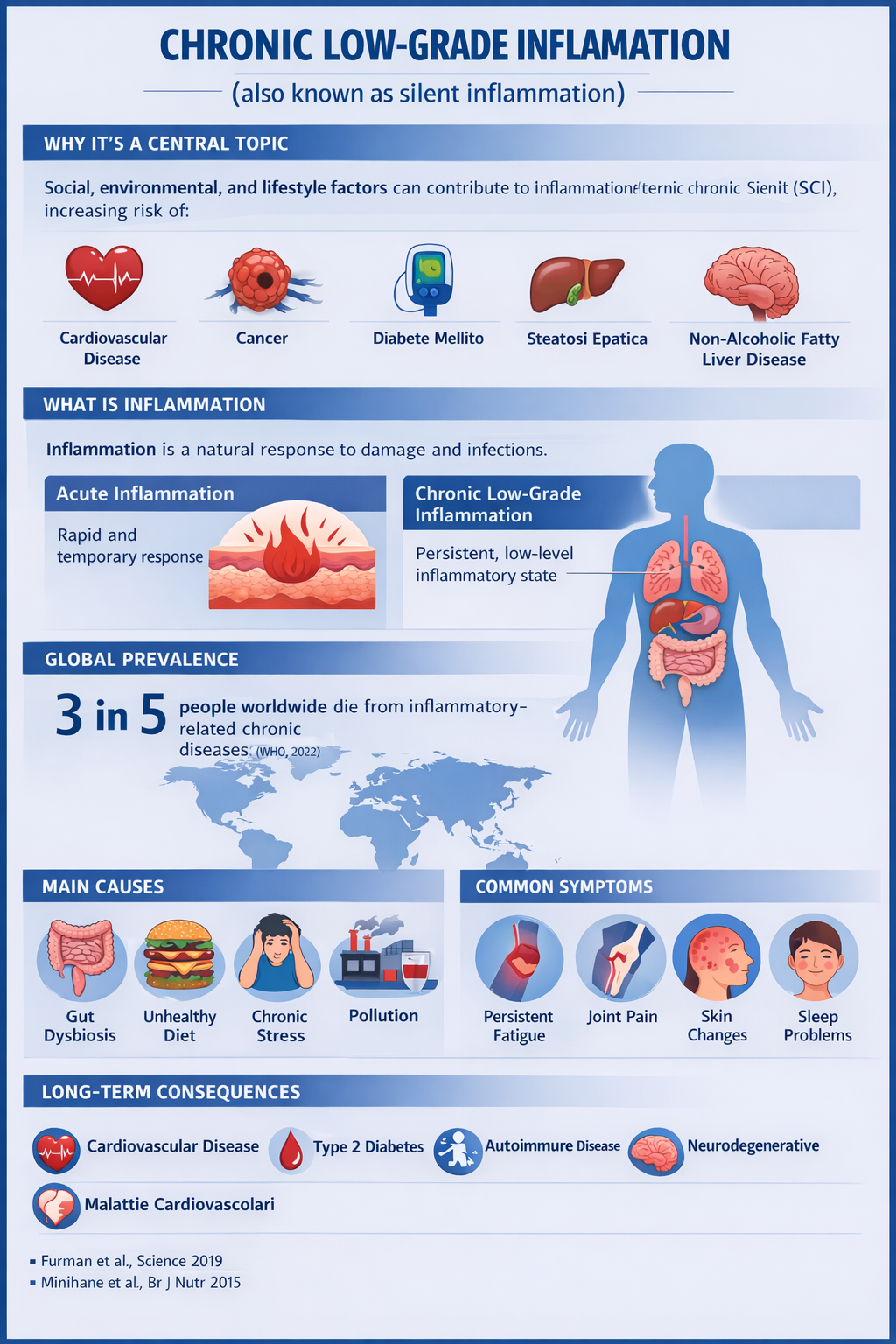
Chronic low-grade inflammation refers to a mild but persistent inflammatory condition of the body, often not very evident or scarcely perceived. Unlike acute inflammation — which is intense, visible, and temporary (as in the case of an infection, injury, or illness) — this form is more silent and may persist over time. In recent years, numerous studies have highlighted how this inflammatory state may contribute to the development or worsening of several metabolic and immune conditions.
Introduction
The proposed diet consists of a set of dietary guidelines and practices aimed at maintaining the intestinal microbiota in balance and promoting the best possible functioning of the immune system.
To achieve this goal, it is useful to reduce or eliminate factors that may alter the balance of the intestinal microbiota and interfere with the efficiency of the immune system.
The microbiota is naturally dynamic: a certain variability is physiological and may depend, for example, on changes in diet, lifestyle, or environment. In response to these variations, the microbiota may adapt physiologically or develop less favorable responses.
Not all variations in the microbiota are therefore negative. However, when these changes lead to persistent imbalances in the intestinal ecosystem, they may promote conditions of microbiota alteration and contribute to the onset of chronic low-grade inflammation.
Reducing this condition is therefore one of the main objectives of the pathway.
Even in the presence of ongoing diseases, adopting dietary and behavioral recommendations that help reduce chronic low-grade inflammation may contribute to preventing further worsening of the clinical condition and to promoting a better overall balance of the organism.
The diet should also be accompanied by some lifestyle guidelines, particularly regarding:
stress and anxiety management
1. regular physical activity
2. balanced lifestyle habits
This aspect is far from marginal. Numerous studies on the gut–brain axis have in fact highlighted a close bidirectional relationship between the nervous system, the intestine, and the microbiota.
Consequently, prolonged stress conditions may negatively influence intestinal balance and may partially or completely compromise the positive effects of a correct and effective diet.
Finally, but no less important, it should be remembered that the great variability of individual psychophysical conditions and the heterogeneity of responses to therapies, treatments, and dietary regimens often require careful personalization of the diet, possibly supported by one’s physician or a specialist.
It should be emphasized from the outset that:
In a truly healthy subject*, the immune system and the organs responsible for regulating homeostasis are physiologically able to maintain the state of health and defend the organism from external agents, including those of dietary origin. This balance depends on the body’s ability to appropriately modulate inflammatory responses, preserve the integrity of the intestinal barrier, and maintain effective communication between the intestine, the immune system, and the nervous system.
The method: what to avoid and why
-
Consuming too much food: the stomach should be able to work (digest) as efficiently as possible. It is better to eat several times rather than having one large meal. The most recent scientific literature suggests that the presence of food that is not completely digested in the intestinal lumen may contribute, in specific contexts [1], to processes of chronic low-grade inflammation and to increased intestinal permeability.
By “specific contexts” we mean the coexistence of an inefficient gastric barrier (hypochlorhydria), slowed intestinal transit (stasis), and altered intestinal permeability (leaky gut), conditions that can transform undigested food residues into pro-inflammatory stimuli for the immune system. -
Meals composed of many different dishes [2]: the simpler the composition of a meal, the easier gastric digestion will be. A significant presence of fats [2.1] may slow the passage of food to the intestine, prolonging digestion and potentially causing sensations of heaviness and bloating. Simple sugars are digested very quickly, usually in the small intestine. However, if they are eaten after a complete meal (perhaps rich in proteins and fiber), they remain “trapped” in the stomach [2.3] while waiting for the rest of the food to be processed and may ferment [3].
-
Industrial food products [4]: as little as possible; they contain additives which, if consumed individually only occasionally, do not usually cause problems but, when accumulated together, may have a more or less marked pro-inflammatory action depending on the individual’s health status. In summary, it is not necessary to rigidly eliminate every food containing additives, but favoring a diet based on minimally processed foods reduces overall exposure to mixtures of additives and represents a simple, safe, and potentially beneficial strategy for intestinal and systemic health.
-
Industrial beverages: as little as possible; they generally contain large amounts of sugar, sweeteners, and additives.
-
Foods for people with celiac disease: as little as possible when there is no real medical necessity. Many industrial gluten-free products may contain high amounts of sugars, fats, and additives, and often have a lower fiber content than traditional products. For this reason, it is preferable to limit their consumption when not strictly necessary. It should also be remembered that the additives contained in these products, when combined, may have a pro-inflammatory effect depending on the individual’s health condition.
-
Wine/beer: with great moderation, because alcohol may interfere with liver metabolism, increase caloric intake, and, if consumed frequently, promote inflammatory processes and alterations of intestinal balance.
-
Spirits: avoid except in occasional situations.
-
Coffee: yes, in amounts compatible with individual tolerance to caffeine, but with attention to the overall sugar content that may accompany it.
-
Spices: yes, favoring those with digestive and antioxidant properties (turmeric, ginger, cinnamon, cumin) and using more irritating ones (black pepper, chili pepper) more moderately.
-
Fried foods: in moderation because frying increases the caloric content of foods and may produce oxidized compounds and irritating substances that, if consumed frequently, may promote inflammatory processes and make digestion more difficult.
-
Fiber: essential. Preferably 3–4 times per day. Fiber represents the main and most important source of nourishment for the microbiota: through it the microbiota produces short-chain fatty acids (butyrate, acetate, propionate) that are beneficial for intestinal health.
-
Processed meats: sparingly, because they generally contain high amounts of salt, preservatives (nitrites and nitrates), and fats—elements which, if consumed frequently, may promote inflammatory processes and metabolic imbalances.
-
Cheese: yes, in amounts compatible with the individual (limited if intolerant to lactose or casein). They should not be completely eliminated when well tolerated, because they represent a good source of proteins, calcium, and other micronutrients useful for the body. It is nevertheless preferable to favor simple, good-quality cheeses consumed in moderation.
-
Sweets: in amounts compatible with the individual. If there are problems with sugars (for weight or blood glucose), they should be consumed in appropriate quantities to avoid imbalances. However, it should not be forgotten that they can also represent a compensatory source of pleasure in many situations of stress or anxiety: moderation yes, but without eliminating them completely.
-
Gluten [5][5.1]: if possible, choose whole or semi-whole wheat pasta; bread: preferably semi-whole or whole made from durum wheat or einkorn/emmer varieties. Soft wheat contains a component of gluten that is very difficult to digest (33mer). Whenever possible, include products made with grains whose gluten is less strong and more tolerable (many ancient grains have these characteristics).
-
Non-celiac gluten sensitivity (NCGS). This type of intolerance is “dose-dependent.” Once it has been established that a person is intolerant but not celiac, it is necessary to identify the quantity that can be tolerated without causing problems. In these cases, products made with grains whose gluten is less tenacious and more tolerable (many ancient grains have these characteristics) may help manage the issue better. It should also be emphasized that many products for people with celiac disease contain several additives: regarding this aspect, see what was stated in point 3 and note [4].
-
Water: drink regularly during the day in adequate quantities. Water is essential for the proper functioning of metabolism, digestion, and waste elimination processes. (Doctors keep reminding us… 1.5–2 liters…)
-
Green tea: because it contains polyphenols and antioxidant substances that may contribute to cellular protection and metabolic balance.
-
Medications: only when truly necessary and under medical prescription.
-
Supplements: to be used after consulting a specialist in order to define a “personalized” intake based on the existing disorder or condition. In addition, many supplements have not been sufficiently tested on large and well-characterized populations.
Specific behaviors:
-
Engage in physical activity, even at a moderate level.
-
If working, try to avoid situations where work leads to excessive stress.
-
If in the post-working phase of life, engage in activities that require concentration and, if possible, creativity. Developing projects is highly beneficial for keeping cognitive functions active.
-
Do not smoke.
-
With your physician, define the routine general check-ups necessary for proper monitoring of your health, in addition to specific examinations for already diagnosed medical conditions.
*It is also important to clarify that the concept of a “healthy subject” does not simply coincide with the absence of clinically diagnosed diseases. In a more rigorous physiological sense, a person can be defined as truly healthy when they do not present ongoing diseases and are not in a state of chronic low-grade inflammation. This distinction is far from marginal, since in clinical practice the term “healthy” is often used in a reductive sense, coinciding only with the absence of formal diagnoses.
Notes:
[1] Undigested food
Low-grade inflammation is not caused by food itself, but by the disruption of the balance between digestion, microbiota, and the intestinal barrier. In particular:
1. Enzymatic and acid failure: If the stomach (due to stress or medications) does not break proteins down into small amino acids, long peptide chains remain that the body may mistake for threats.
2. Biochemical transformation: Undigested residues, when stagnating, undergo processes of putrefaction (proteins) or excessive fermentation (sugars), producing toxic metabolites (ammonia, phenols, gases) that irritate the intestinal mucosa.
3. The immune breach: In the presence of a “permeable” intestinal mucosa, these macromolecules and toxins cross the cellular wall and come into direct contact with the immune system, keeping it in a constant state of alert (release of inflammatory cytokines).
[2] Simplicity and enzymatic “load”
Each macronutrient (carbohydrates, proteins, fats) requires different enzymes and breakdown times. When we mix too many different foods:
-
The stomach must manage a complex chemical mixture.
-
The body struggles to optimize gastric pH for each food.
Result: A faster and “cleaner” digestion occurs when meals consist of a few well-combined ingredients.
[2.1] The role of fats
Fats are the slowest nutrients to digest. Their presence sends hormonal signals (such as cholecystokinin) that tell the stomach to slow the emptying toward the duodenum.
The positive side: They provide a prolonged sense of satiety.
The negative side: If the meal is excessively fatty, food stagnates in the stomach. This process of stagnation or fermentation is what causes the sensation of a “brick in the stomach” and abdominal bloating.
[2.3] Tips for a balanced but light meal
To avoid heaviness without giving up taste, you could follow these small precautions:
-
Prefer simple cooking methods: steaming, grilling, or baking rather than frying or prolonged sautéing.
-
Limit different protein sources: avoid mixing eggs, cheese, and meat in the same meal.
-
Add fats raw: use extra virgin olive oil at the end of cooking to preserve its properties and facilitate digestion.
In summary
The fewer “obstacles” we give our digestive system in the form of complex combinations and heavy fats, the more energy we will have available after a meal instead of feeling sleepy and bloated.
[3] Sugars
While fats slow digestion for reasons of “biochemical management” (the stomach closes the valve to take more time), simple sugars consumed at the end of a meal (here quantity plays an important role) create a sort of digestive “queue” in the stomach.
3.1. The “plug” effect and fermentation
Simple sugars are digested very quickly, usually in the small intestine. If they are consumed after a complete meal (perhaps rich in proteins and fiber), they remain “trapped” in the stomach while waiting for the rest of the food to be processed.
Consequence: In that warm and humid environment, sugars begin to ferment.
Result: Gas production, immediate abdominal bloating, and a sensation of acidity.
3.2. Fluid attraction (Osmosis)
Sugars are “osmotic” substances, meaning they attract water into the stomach and intestines in order to be diluted.
This influx of fluids can cause a sensation of abdominal distension and, in some cases, cramps or accelerated intestinal transit (not necessarily in a beneficial sense).
3.3. The impact on insulin
Unlike fats, which do not significantly stimulate insulin, a dessert at the end of a meal (again, quantity plays an important role) may cause a significant glycemic spike.
If the preceding meal was already rich in carbohydrates (pasta or bread), the dessert becomes the “last drop that makes the cup overflow.”
This spike is often followed by a crash (reactive hypoglycemia) that makes you feel tired and lacking energy shortly after eating.
|
Characteristic |
High Fat |
Sugars (Sweets) |
|
Main action |
Slow gastric emptying. |
Ferment while waiting to be digested. |
|
Sensation |
Heaviness, “stone in the stomach”. |
Bloating, gas in the abdomen, drowsiness. |
|
Hormonal effect |
Prolonged feeling of satiety. |
Insulin spike followed by fatigue. |
3.4. Fermentation in the stomach