Highlights:
1️⃣ Einkorn (Triticum monococcum) possesses a predominantly visco-colloidal dough matrix, due to the greater prevalence of gliadins compared to polymeric glutenins, which results in doughs that are less elastic and more viscous than those of modern wheat.
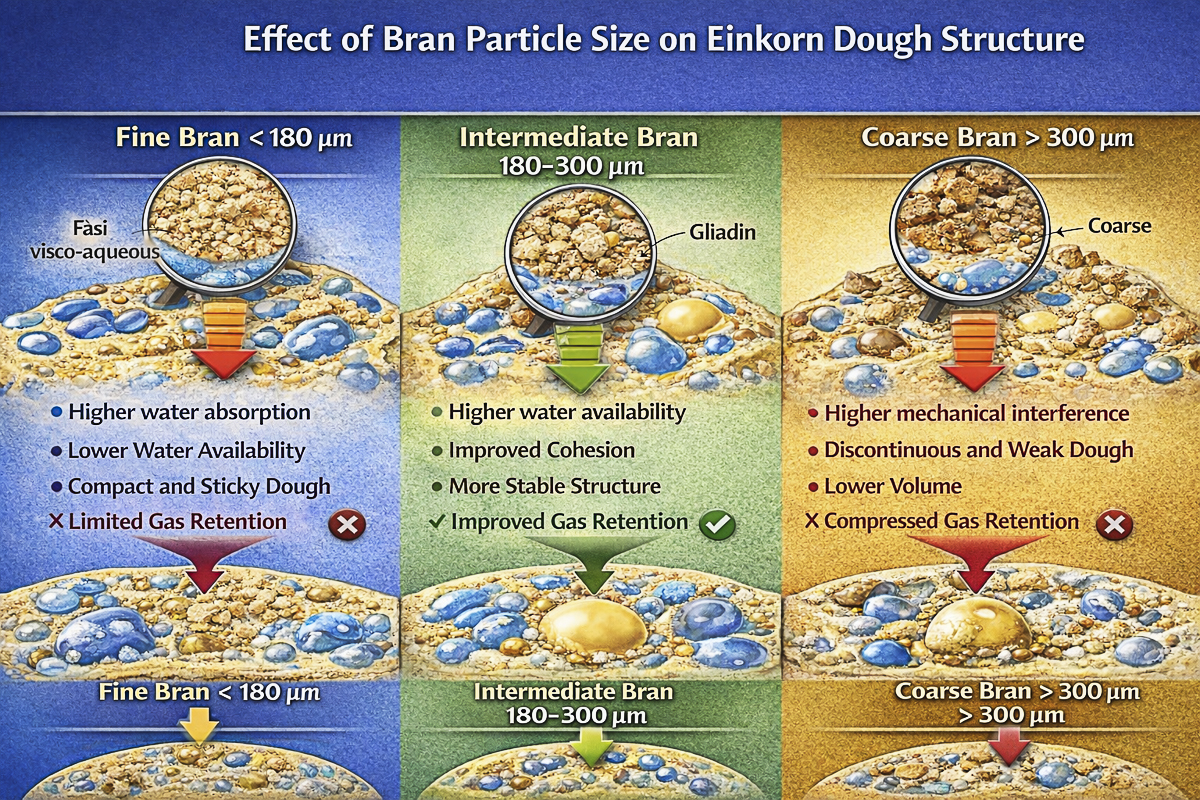
2️⃣ Bran particle size represents a crucial technological parameter in wholegrain flours, influencing water absorption, dough cohesion and fermentation stability.
3️⃣ In einkorn, an intermediate bran particle size may have a structuring effect on the dough, acting as a colloidal filler within the matrix and contributing to the stabilization of gas bubbles during fermentation.
4️⃣ Genetic variability among einkorn genotypes significantly influences technological quality, with relevant differences in dough behavior, bread volume and final aromatic profile.
5️⃣ Some einkorn lines show relatively lower gluten immunogenicity compared to hexaploid wheats, although they are not suitable for the diet of celiac patients. However, they may be useful for certain individuals (see end of chapter 11).
1. Introduction
Einkorn wheat (Triticum monococcum) represents one of the oldest wheat species cultivated by humans and possesses technological characteristics that differ significantly from those of modern wheats. In particular, the rheological properties of einkorn flours differ substantially from those of modern bread wheat, especially with regard to the structure and behavior of the gluten matrix.
The protein composition of einkorn is characterized by a relative predominance of gliadins (including γ-gliadins) and by a lower quantity and quality of polymeric glutenins. Gliadins mainly contribute to the viscous properties of the dough, while polymeric glutenins are responsible for elastic properties and for the formation of a stable three-dimensional gluten network.
This specific protein composition results in a rheological system in einkorn that behaves predominantly as a pasty-viscous system rather than an elastic one (Figure 1). Consequently, doughs obtained from einkorn flours are generally less elastic, more viscous and have a limited capacity to retain gas during fermentation.

Scientific references
Wieser, H. (2007). Chemistry of gluten proteins. Food Microbiology. DOI: 10.1016/j.fm.2006.07.004
Abdel-Aal, E.-S. M. et al. (1998). Genetic and environmental effects on gluten proteins of einkorn wheat. Journal of Cereal Science. DOI: 10.1006/jcrs.1997.0143
2. Role of Bran in Dough: General Concepts
Bran represents a fundamental component of wholegrain flours and can significantly influence the rheological properties of dough and the quality of the final product. The effect of bran on dough is generally attributed to two main mechanisms: interaction with water and mechanical interference with the dough structure.
2.1 Water absorption effect
Bran particles possess a remarkable capacity to absorb water due to their high content of dietary fiber, particularly arabinoxylans and cellulose. As the specific surface area of bran particles increases, their capacity to bind water also increases.
✅ This phenomenon results in a reduction of water available for other dough components, particularly starch and gluten proteins. Consequently, the distribution of water in the dough can significantly modify the formation and stability of the protein matrix.
2.2 Mechanical effect of bran particles
In addition to the water-related effect, bran can exert a mechanical effect on the dough structure. Bran particles of large size may act as discontinuous elements within the dough matrix, interfering with the continuity of the gluten network.
In modern wheats, characterized by a relatively strong and elastic gluten network, coarse bran particles can physically interrupt the protein network, resulting in a reduced ability of the dough to retain gas and, consequently, a decrease in final bread volume.
References
Noort, M. W. J. et al. (2010). The effect of particle size of wheat bran on bread quality. Journal of Cereal Science. DOI: 10.1016/j.jcs.2010.03.003
Hemdane, S. et al. (2016). Wheat bran in bread making: A critical review. Food Chemistry. DOI: 10.1016/j.foodchem.2015.09.092
3. Effect of Bran Particle Size on Dough Properties
The size of bran particles represents a particularly important technological parameter, as it influences both water absorption capacity and mechanical interaction with the dough structure.
3.1 Fine bran
Fine bran presents a high specific surface area. This results in a greater capacity to absorb water compared to larger particles.
In the presence of fine bran, the following are generally observed:
1️⃣ lower water availability for proteins and starch
2️⃣ higher water absorption by bran
3️⃣ more homogeneous distribution of particles in the dough.
From a technological point of view, these effects may lead to the formation of more viscous and compact doughs, with a more limited but generally more uniform development of dough structure.
3.2 Coarse bran
Bran with larger particle size presents a lower specific surface area and therefore tends to absorb less water during the initial phases of mixing.
However, larger particles may exert a stronger mechanical effect on the dough structure. In modern wheats this phenomenon may cause discontinuities in the gluten network, resulting in reduced dough stability and lower final bread volume.
4. Technological Specificity of Einkorn
In the case of einkorn, the effect of bran must be interpreted in light of the specific characteristics of its protein matrix.
As previously described, the gluten network of einkorn is generally weaker than that of modern wheats and does not form an equally developed continuous elastic structure. Dough behavior is dominated more by viscosity and colloidal cohesion phenomena rather than by a well-organized elastic gluten network.
✅ In this technological context, bran does not necessarily act as an element that breaks a strong gluten network, as occurs in modern bread wheat. However, it may still interfere with dough cohesion or contribute to the stabilization of the overall structure of the system.
References
Hidalgo, A. & Brandolini, A. (2014). Nutritional properties of einkorn wheat. Journal of the Science of Food and Agriculture. DOI: 10.1002/jsfa.6382
Brandolini, A. et al. (2008). Technological quality of einkorn wheat. Journal of Cereal Science. DOI: 10.1016/j.jcs.2008.01.001