(Low-grade chronic inflammation is not a disease in the strict sense, but a persistent biological state that promotes the development of numerous chronic conditions. This document proposes an integrated approach aimed at modulating it through lifestyle.)
Furthermore:
In the absence of unique and definitive solutions, the most rational strategy for reducing low-grade chronic inflammation consists of adopting a lifestyle model that minimizes exposure to potentially pro-inflammatory factors* and promotes protective ones.
The importance of low-grade chronic inflammation
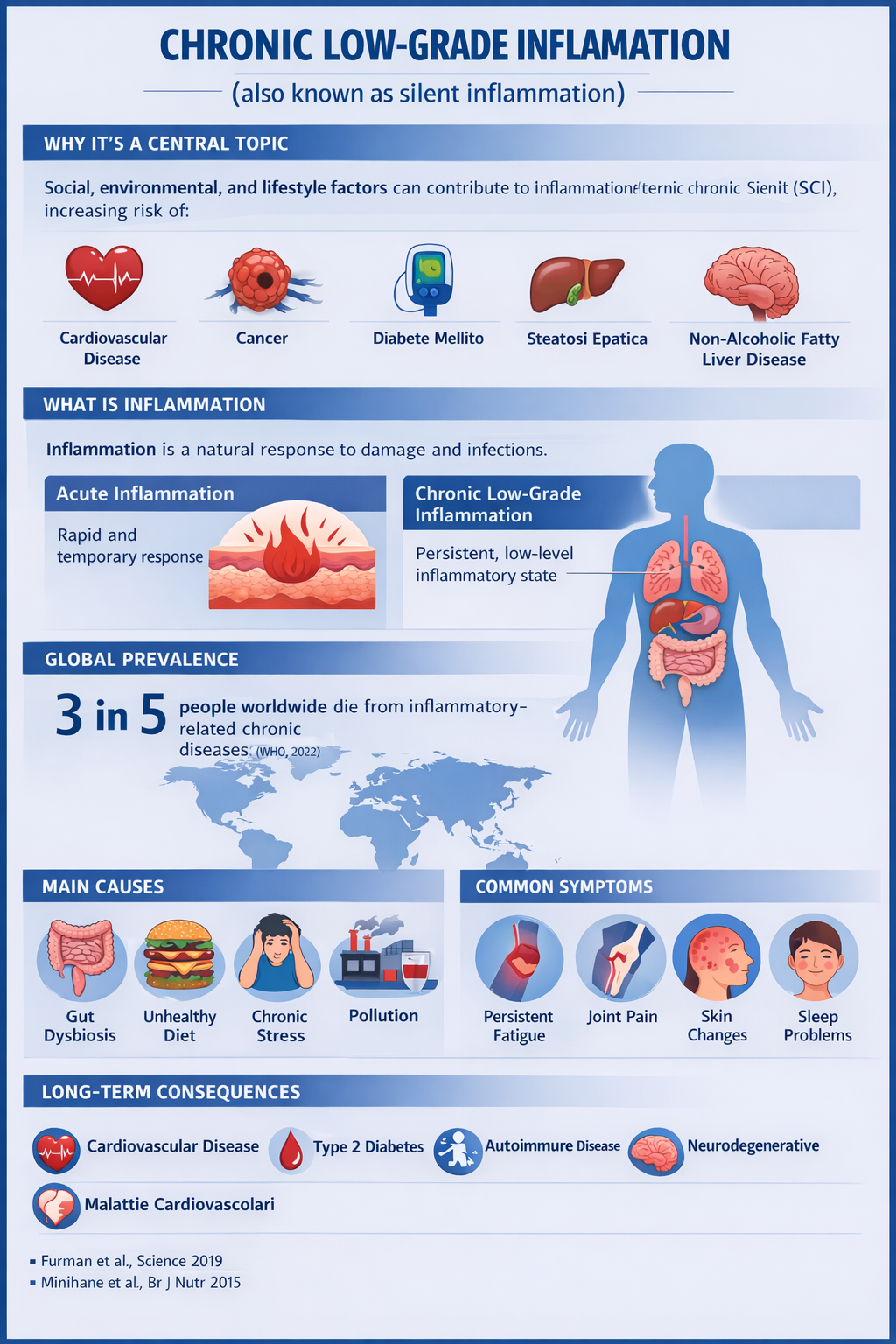
Although intermittent increases in inflammation are essential for survival during physical injury and infections, recent research has revealed that certain social, environmental, and lifestyle-related factors can promote chronic systemic inflammation, particularly low-grade chronic inflammation (LGCI), which in turn may lead to several diseases that, taken together, represent the leading causes of disability and mortality worldwide, such as cardiovascular diseases, cancer, diabetes mellitus, chronic kidney disease, non-alcoholic fatty liver disease, and autoimmune and neurodegenerative diseases.
(see article: https://glutenlight.eu/2025/08/21/infiammazione-cronica-basso-grado/)
This type of inflammation has multiple triggers:
Gut dysbiosis:
Alteration of the intestinal bacterial flora, which may be caused by an unbalanced diet, excessive use of antibiotics, or other toxic substances.
Unhealthy diet:
Excessive consumption of processed foods rich in refined sugars and saturated fats, which can promote inflammation.
Stress:
Chronic stress can negatively affect the immune system and increase susceptibility to inflammation.
Environmental pollution and toxins:
Exposure to chemicals present in the environment or in foods can contribute to oxidative stress and inflammation.
Smoking and alcohol:
These factors can aggravate oxidative stress and damage cells, promoting inflammation.
(see article: Oxidative stress)
Among the triggers, drug use is not mentioned because drugs are always and in any case considered to be avoided.
The integrated approach must necessarily involve the individual in all aspects of life. This is the central point: a lifestyle model must be “built.” And this model must be personalized.
Another consideration concerns the individual’s general health status, which should primarily remain “healthy,” that is, free from diseases, trauma, wounds, etc., which activate acute inflammation.
It is important to emphasize that, in the presence of acute inflammation, the biological markers used to assess low-grade chronic inflammation appear elevated, making it difficult to distinguish between the two phenomena and potentially masking improvements in LGCI.
With these clarifications, we can begin the integrated approach.
1 – Stress management
This is a very important factor, considering emerging scientific evidence regarding the gut–brain axis, a bidirectional communication system through which psychological stress, emotions, and mental states influence intestinal motility, barrier permeability, and microbiota composition, and vice versa. Alterations of this axis can promote inflammation, digestive disorders, and metabolic imbalances.
Stress should be managed either independently using available techniques or, if not possible, with the help of a psychologist.
2 – Environmental pollution (air, water, etc.)
It goes without saying that the more we can avoid it, the better. This factor is relevant to oxidative stress.
3 – Nutrition: here we can do a lot
Important point:
Diet must be strictly correlated with age, type of activity, eating habits, and general health status.
Foods to avoid
-
Industrial foods: contain additives that, if taken occasionally and individually, do not cause problems, but if combined together may exert a more or less marked pro-inflammatory action depending on the subject’s health status [A].
-
Industrial beverages: generally contain many sugars/sweeteners/additives.
-
Many gluten-free products (especially industrial ones) are highly processed and contain additives (often many) that, if taken occasionally and individually, do not cause problems, but if combined together may have a pro-inflammatory action depending on the subject’s health status.
Foods to consume in moderation
-
Wine/beer: in moderation.
-
Alcoholic beverages: on rare occasions (spirits: NO).
-
Coffee: in moderation.
-
Processed meats: with great moderation.
-
Sweets: in moderation. If there are issues with sugars (weight or glycemia), they must be consumed in appropriate amounts to avoid problems.
-
Cheeses: with great moderation and in amounts compatible with the individual (if lactose/casein intolerant).
-
Spices: in moderation.
-
Fats: less trans (hydrogenated) fats and, to a lesser extent, excess saturated fats, and more extra virgin olive oil (oleic acid).
-
Refined sugars: the less, the better. It should also be remembered that frequent spikes in glucose and insulin stimulate the production of pro-inflammatory cytokines
-
Gluten: in moderation. If possible, whole-grain/partially whole-grain pasta; bread: if possible, whole durum wheat/spelt. Soft wheat contains a gluten component that is very difficult to digest (33-mer). Considering the relationship between gluten strength and digestibility, products made with grains that have less tenacious gluten should be preferred. Among “ancient grains,” many with this characteristic can be found (in reality, even among modern grains there are cultivars with less tenacious gluten, often used for pastries rather than bread). These should be preferred.
-
Those who are gluten intolerant but not celiac, considering that this intolerance is “dose-dependent,” can, with the help of a physician, try to identify the threshold (quantity) that does not cause problems. Grains with less tenacious gluten facilitate the possibility of consuming products made from them. Further reading: Difference between ancient and modern grains (published separately)
Foods to consume in abundance
-
Fiber (compatible with any intestinal issues): 3–4 times per day.
-
Fruit (depending on any sugar-related issues: blood sugar and/or weight).
-
Green tea: an excellent ally. Green tea is considered a powerful ally against chronic inflammation thanks to its rich composition of bioactive compounds that act on multiple fronts of the body [D].
It’s worth noting the essential contribution of water to maintaining effective hydration. The lymphatic system is a sort of drainage network for chronic inflammation.
But it only works well if there’s enough water. If you drink too little, lymph stagnates, toxins remain in the tissues, and “background” inflammation increases. See: The Role of Water in Reducing Low-Grade Inflammation
4 – Eating behaviors
Nutrition rests on two main pillars: quantity and quality.
The quantity of food consumed should be what is necessary for physiological functions plus what is required for activities performed. This simple principle would greatly help maintain a correct and healthy weight. Not easy for two simple reasons: the first is “gluttony,” the second is that the “full/satiated” mechanism is delayed compared to actual fullness; the sensation of satiety does not coincide with real stomach filling but arrives later. Already 50 years ago, family doctors suggested leaving the table with a slight desire for more food.
Quality: it goes without saying that the more genuine and “clean” (i.e., free of toxic substances) foods are, the better.
The following should also be considered a general framework because, as stated, it must be “designed around the individual.”
A – Avoid consuming too much food in a single meal
The stomach should be allowed to work (digest) optimally. It is often preferable to eat more frequently rather than having a single very large meal. Ideally, one should finish eating and “not feel the stomach,” with the result of no postprandial “fog.”
Further reading: Why smaller, distributed meals work better (published separately)
Food that is not completely digested, in healthy individuals*, is subsequently processed in the intestine and then expelled. However, if the gastrointestinal system is compromised or altered, the passage of inadequately digested substrates into the intestine may promote bacterial fermentation and be pro-inflammatory. (https://glutenlight.eu/2025/06/12/cibo-non-digerito-e-infiammazione-intestinale/)
Not only the stomach, but also and especially the intestine must be able to function optimally and continue digesting food in order to make it absorbable. [B] [C]
*The critical point here is: does a truly healthy individual still exist?
B – Avoid mixing very different foods
The stomach works in an acidic environment, where pepsin digests proteins (further digested in the intestine by trypsin and other enzymes). Sugars begin digestion in the mouth (ptyalin) and are then mainly digested in the intestine (pancreatic amylase). Some clarifications are necessary:
Carbohydrates and proteins in the stomach generally do not cause problems.
A pasta course followed by fish, meat, cheese, and perhaps vegetables, in amounts appropriate to one’s digestive capacity, does not cause problems.
If the second course is very fatty, gastric digestion slows and, depending on quantity, gastric emptying may be delayed, with possible passage of incompletely digested food into the intestine.
The situation is different if a dessert is included.
Here we face a significant amount of simple sugars, not complex carbohydrates (pasta, for example, is mainly starch, and only part of it is transformed into sugars already in the mouth; therefore, mainly starch reaches the stomach).
Sugars are not digested in the stomach except to a negligible extent:
“The stomach has a highly acidic environment that prevents fermentation there; the undigested sugars travel to the small intestine and large intestine, where they are fermented by the gut bacteria.”
Dessert at the end of a meal (intended as a moderate portion) does not cause problems in a healthy person (who today is relatively rare), but it makes digestion more difficult for many people, not only because of possible subsequent intestinal effects, but also due to the sensation of heaviness that may appear.
It should be clarified that this is not a dogma: there are people who digest practically everything without difficulty—we are all different.
Age also plays a fundamental role. Elderly individuals tend to feel better when meals are simpler.
Further reading: Sugars and proteins in gastric digestion (published separately)
Important point
In the presence of diet-related pathologies, the intervention of a specialist (dietitian or nutritionist) is strictly necessary.
5 – Specific behaviors
-
Engage in physical activity, even moderately.
-
If working, avoid work that leads to stress. Stress must be managed, otherwise it becomes a cause of low-grade chronic inflammation.
-
If overweight, weight must be reduced.
-
After work, engage in activities that require concentration and, if possible, creativity. Developing projects is highly useful for keeping brain functions active.
6 – Medical evaluations
With one’s physician, define the routine general tests necessary for good monitoring of one’s health, as well as specific tests for any conditions.
Final Summary
We must build a personalized lifestyle model for reducing low-grade chronic inflammation.
In a healthy person, a meal containing proteins and sugars in moderate amounts does not create problems. The association becomes potentially problematic when sugars are highly concentrated, especially in liquid form and in large quantities. In individuals with a sensitive or altered gastrointestinal system, even moderate portions (such as dessert at the end of a meal) may cause digestive discomfort.
The integrated approach to reducing low-grade chronic inflammation is based on the available scientific evidence reported in the bibliography section.
Since many studies show significant associations without demonstrating an absolute causal relationship, a precautionary principle is adopted: reduce or eliminate, where possible, potentially harmful factors, favoring choices with low biological risk.