(Role of yeasts, enzymes, lactic acid bacteria, and intestinal microbiota)
General Index
-
Structure of gluten
-
What gluten hydrolysis is
-
The role of baker’s yeast
-
Enzymes naturally present in flour
-
Biochemical processes in dough
-
Short fermentation (4 hours) with baker’s yeast
-
Prolonged fermentation (12 hours) with baker’s yeast
-
Why sourdough is different
-
Microbiology of sourdough
-
Effects of lactic fermentation
-
Degradation of immunogenic gluten peptides
-
Reduction of FODMAPs
-
Role of temperature in fermentation
-
Role of dough hydration
-
Difference between leavening and maturation
-
Why too much hydrolysis worsens structure
-
The role of flour strength (W)
-
How much gluten is actually hydrolyzed in studies
-
Human digestion of gluten
-
Actual digestibility and sensation of lightness
-
The myth of 48–72 hour maturation
-
Why some pizzas cause bloating
-
Role of the intestinal microbiota
-
Gluten or fructans: what causes the symptoms
-
Reduction of FODMAPs with sourdough
-
Individual variability in digestion
-
General scheme of the biochemical processes of dough
-
Main chemical reactions during fermentation
-
Evolution of dough structure during fermentation
-
Percentages of gluten degradation in experimental studies
-
General conclusions
-
Bibliography
1 . Structure of gluten [1][3]
Gluten is a protein complex present in wheat that forms when flour is hydrated and subjected to mixing. The main proteins involved are gliadins and glutenins, which belong to the class of wheat storage prolamins.
Gliadins are monomeric proteins soluble in hydroalcoholic solutions and are mainly responsible for the viscosity and extensibility of the dough.
Glutenins, on the other hand, are polymeric proteins composed of high- and low-molecular-weight subunits (HMW-GS and LMW-GS) linked together by disulfide bonds. These proteins give the dough elasticity and strength.
During mixing, the interactions between gliadins and glutenins lead to the formation of a three-dimensional viscoelastic protein network capable of retaining the gases produced during fermentation and responsible for the structure of baked products [1].
A fundamental role in gluten structure is played by GMP (Glutenin Macropolymer), large aggregates of high-molecular-weight glutenins that constitute the insoluble fraction of gluten. GMP represent the structural backbone of the gluten network and are strongly correlated with flour strength, dough tenacity, and baking quality [3].
The stability of the gluten network mainly depends on several intermolecular interactions, including:
• disulfide bonds between glutenin subunits
• hydrogen bonds between polar groups of proteins
• hydrophobic interactions between nonpolar regions of protein chains.
2 . What gluten hydrolysis is [2]
Hydrolysis consists of the cleavage of peptide bonds in proteins with the formation of smaller peptides.
In the case of gluten, the process is catalyzed by proteolytic enzymes (proteases), which use a water molecule to break the peptide bond of the protein chain.
In general terms, the reaction can be schematized as:
protein + H₂O —(protease)→ smaller peptides
Hydrolysis modifies the structure of the gluten network and the rheological properties of the dough. In particular:
When hydrolysis is moderate:
• dough extensibility increases
• workability improves
When hydrolysis is excessive:
• the gluten network weakens
• the ability to retain fermentation gases is reduced [2].
3. The role of baker’s yeast [4]
Saccharomyces cerevisiae is responsible for alcoholic fermentation.
Metabolic reaction: glucose → CO₂ + ethanol.
Effects:
• dough growth
• aromatic development.
Yeast produces limited amounts of proteases and therefore does not significantly contribute to gluten degradation [4].
4. Enzymes naturally present in flour [4][5]
Flour contains several endogenous enzymes that play a fundamental role in the biochemical transformations occurring during mixing and fermentation. These enzymes contribute to the controlled degradation of the main macromolecules of flour, particularly starches and proteins.
Among the most important enzymes are amylases, which catalyze the degradation of starch. In particular, starch is progressively hydrolyzed according to the sequence:
starch → maltose → glucose
The simple sugars produced represent an energy source for yeasts, which use them in fermentative metabolism to produce carbon dioxide (CO₂) and other metabolites involved in dough development [4].
Another important group of enzymes is represented by proteases, which act on gluten proteins. These enzymes progressively hydrolyze protein chains according to the process:
gluten → polypeptides → peptides
This partial degradation of proteins modifies the structure of the gluten network, making the dough more extensible and workable.
Taken together, the reactions catalyzed by amylases and proteases contribute to the processes of enzymatic maturation of the dough, which influence the structure, fermentability, and final characteristics of the baked product [5].
5. Biochemical processes in dough [4][5]
When flour and water are mixed, a series of physical, chemical, and microbiological processes are activated that determine the evolution of the dough over time. In particular, three main interconnected systems are activated:
a. formation of the gluten network
b. enzymatic activity of the flour
c. microbial fermentation
Hydration of the flour allows gluten proteins (gliadins and glutenins) to absorb water and interact with one another, progressively forming a three-dimensional protein network that gives the dough elasticity and extensibility.
At the same time, the enzymes naturally present in flour, such as amylases and proteases, are activated and begin to degrade starches and proteins respectively, contributing to the processes of dough maturation.
Finally, the yeasts and any lactic acid bacteria present in the dough metabolize the available sugars, producing carbon dioxide (CO₂) and other metabolites, responsible for the increase in dough volume and the development of aromatic compounds.
Under normal conditions, the processes in the dough follow a typical sequence:
a. hydration of proteins and initial formation of the gluten network
b. activity of amylases, with starch degradation and production of fermentable sugars
c. yeast fermentation, with production of CO₂
d. enzymatic maturation, which progressively modifies the structure of starches and proteins.
These processes occur simultaneously and interdependently, influencing the structure of the dough, its workability, and the characteristics of the final product.
6. Short fermentation (about 4 hours) [6]
During a short fermentation, as in baking processes with reduced times, the predominant activity is that of yeasts, which metabolize the available sugars producing carbon dioxide (CO₂) and ethanol. The CO₂ remains trapped in the gluten network and causes the increase in dough volume.
Under these conditions, the time available for enzymatic maturation processes is limited. Consequently, the enzymes present in flour, particularly proteases, have little time to act on gluten proteins.
The result is that:
a. the hydrolysis of gluten proteins remains very limited
b. the structure of the gluten network undergoes few modifications
c. the dough maintains a relatively compact and little-modified protein structure [6].
7. Prolonged fermentation (about 12 hours) [5].
During longer fermentations, the dough remains for a longer time under the combined action of enzymes and microorganisms, allowing a more complete development of maturation processes. In particular, the proteases present in flour begin to hydrolyze some chains of gluten proteins, partially breaking peptide bonds. This process leads to a reduction in the rigidity of the gluten network and makes the dough more extensible and workable.
At the same time, amylases continue to degrade starch producing simpler sugars, which can be used by yeasts in the fermentative process. During fermentation a slight decrease in the pH of the dough is also observed, due to the production of organic acids by microorganisms. This lowering of pH can favor the activity of some enzymes, further contributing to the processes of enzymatic maturation of the dough [5].
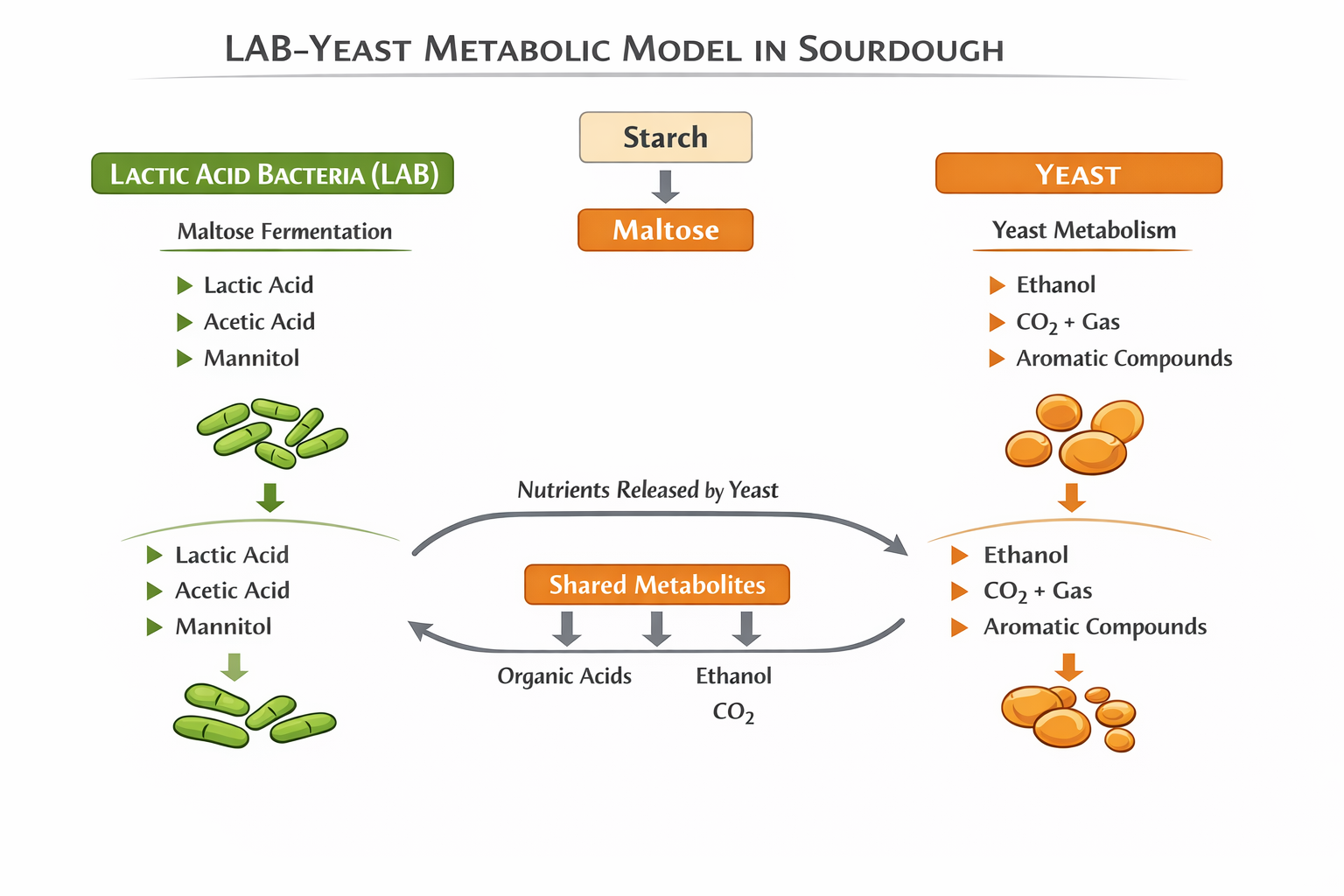
8. Why sourdough is different [5][8]
Sourdough contains:
• yeasts
• lactic acid bacteria.
Lactic acid bacteria produce:
• lactic acid
• acetic acid.
This acidification increases the activity of proteases and the degradation of gluten [5].
Some lactic acid bacteria possess complex proteolytic systems, including enzymes such as prolyl endopeptidases and endopeptidases specific for proline residues, which are able to further degrade proline-rich peptides, including some immunogenic fragments of gluten.
Among the microorganisms most studied for this activity are species belonging to the genera Lactobacillus, Lactiplantibacillus and Lacticaseibacillus, including:
• Lactobacillus sanfranciscensis
• Lactobacillus plantarum (now Lactiplantibacillus plantarum)
• Lactobacillus brevis
• Lactobacillus helveticus
• Lactobacillus paracasei
These microorganisms, often present in sourdough starters, possess proteolytic systems capable of hydrolyzing proline-rich peptides and contributing to the degradation of immunogenic sequences of gliadin, including fragments derived from the 33-mer peptide, reducing their immunogenic potential [8].
9. Microbiology of sourdough
Sourdough hosts a complex and relatively stable microbial community, composed mainly of yeasts and lactic acid bacteria, which coexist in a natural fermentation system. The balance between these microorganisms depends on several factors, including type of flour, dough hydration, temperature, and refreshment method.
Among the yeasts most frequently associated with sourdough are:
• Saccharomyces cerevisiae
• Candida milleri (now often classified as Kazachstania humilis)
These microorganisms are mainly responsible for the production of carbon dioxide (CO₂) through sugar fermentation, contributing to the increase in dough volume.
Alongside yeasts, sourdough contains numerous lactic acid bacteria, including:
• Lactobacillus sanfranciscensis
• Lactobacillus plantarum
• Lactobacillus brevis
Lactic acid bacteria metabolize sugars producing lactic acid and acetic acid, contributing to the development of dough acidity and the formation of aromatic compounds characteristic of natural fermentation [7].
10. Effects of lactic fermentation [5][7]
Lactic fermentation plays an important role in the biochemical evolution of the dough during sourdough fermentation. One of the main effects is the progressive acidification of the dough, due to the production of lactic acid and acetic acid by lactic acid bacteria. The decrease in pH influences several biochemical and technological processes. In particular, acidification can favor greater activity of some enzymes present in flour, such as amylases and proteases, contributing to the processes of enzymatic maturation of the dough. In addition, the presence of lactic acid bacteria and their proteolytic systems can lead to greater degradation of proteins, including a partial hydrolysis of gluten proteins. This process can modify the structure of the protein network, making the dough more extensible and workable. Overall, lactic fermentation contributes not only to the development of acidity, but also to the formation of aromas, the modification of dough structure, and the final characteristics of the baked product.
11. Degradation of immunogenic peptides [8]
During During gastrointestinal digestion, gluten proteins are partially hydrolyzed by digestive enzymes such as pepsin, trypsin, and chymotrypsin. However, due to the high content of proline and glutamine, some peptide sequences are particularly resistant to enzymatic degradation.:
This resistance to digestion is largely due to the high content of proline residues in gluten proteins, which makes many peptide bonds poorly accessible to human digestive enzymes.
Among Among these fragments, some peptides are immunogenic, that is, capable of activating the immune response in subjects affected by celiac disease. One of the most studied is the 33-mer peptide derived from α-gliadin, known for its high resistance to digestion and for the presence of several epitopes recognized by the immune system. Its resistance derives from the high content of proline and glutamine, which makes the peptide poorly susceptible to human digestive enzymes.
In addition to immunogenic peptides, resistant but non-immunogenic peptides are also formed during gluten digestion, which are not capable of activating the typical immune response of celiac disease. These fragments derive from the partial degradation of gluten proteins and can persist in the gastrointestinal tract.
In individuals with alterations in gastrointestinal functionality, such as in the case of a compromised intestinal barrier or microbiota dysbiosis, such peptides may contribute to undesirable biological effects. In particular, some studies suggest that they may interact with the intestinal microbiota, influence the permeability of the epithelial barrier, and modulate, albeit indirectly, some local immune responses.
Even undigested non-immunogenic peptides can persist in the intestinal lumen and participate in microbial fermentation processes or interact with the microbiota. Although they do not activate the typical immune response of celiac disease, their presence may contribute, in predisposed subjects, to the appearance of gastrointestinal symptoms or to a sensation of reduced digestibility.
12. Reduction of FODMAPs and modification of other wheat compounds [9]
Wheat contains fructans, carbohydrates belonging to the category of FODMAPs (Fermentable Oligo-, Di-, Monosaccharides and Polyols). These compounds may be poorly digested in the small intestine and fermented by the intestinal microbiota, causing gastrointestinal symptoms in sensitive individuals. During lactic fermentation, some microorganisms are able to metabolize fructans through enzymes such as fructanases, converting them into simpler sugars that are subsequently transformed into organic acids (mainly lactic acid and acetic acid):
fructans → simple sugars → organic acids
13. Role of temperature [4]
Temperature Temperature is one of the main factors that influence the speed and balance of the processes occurring in dough, particularly microbial fermentation and enzymatic activity.
At higher temperatures, the metabolic activity of yeasts and bacteria increases, resulting in faster fermentation and more rapid production of CO₂. However, fermentation that is too fast can reduce the time available for enzymatic maturation processes, with less degradation of starches and proteins.
Conversely, at lower temperatures fermentation occurs more slowly. This slowing of microbial activity allows the enzymes present in flour (such as amylases and proteases) to act longer on the dough, favoring greater enzymatic maturation.
Consequently:
• high temperatures → fast fermentation and shorter processing times;
• low temperatures → slower fermentation and greater development of maturation processes.
For this reason, modern baking techniques often use controlled low-temperature fermentations, which allow better management of production times and favor the development of structure, aromas, and rheological characteristics of the dough.
14. Role of hydration [5]
Dough hydration, that is, the amount of water present relative to the flour, represents a fundamental factor in the processes that occur during mixing and fermentation.
Water performs several technological and biochemical functions, including:
• favoring the molecular mobility of dough components
• allowing the development of the gluten network through hydration of gluten proteins
• facilitating the activity of enzymes present in flour and produced by microorganisms.
An adequate availability of water allows enzymes, such as amylases and proteases, to act more effectively on starches and proteins, favoring the maturation processes of the dough.
More hydrated doughs therefore tend to show greater enzymatic activity and a more extensible structure, allowing more effective maturation during fermentation. In addition, greater hydration can favor the formation of a more alveolated structure in the final product.
Conversely, doughs with low hydration are generally more compact and limit the mobility of molecules and enzymatic activity, partly reducing the intensity of maturation processes [5].
15. Difference between leavening and maturation [4][5]
In the baking process it is important to distinguish between leavening and maturation, two phenomena that often occur simultaneously but involve different mechanisms.
Leavening Leavening is the biological process through which yeasts metabolize the sugars present in the dough producing carbon dioxide (CO₂) and ethanol. The CO₂ remains trapped in the gluten network, determining the increase in dough volume and the formation of alveolation in the final product.
Maturation, on the other hand, mainly concerns the enzymatic processes that occur in the dough over time. During this phase, enzymes naturally present in flour or produced by microorganisms act on starches and proteins.
In particular:
• amylases degrade starch into simpler sugars, which can be used by yeasts during fermentation;
• proteases partially hydrolyze gluten proteins, modifying the structure of the gluten network.
These processes contribute to making the dough more extensible and workable, as well as influencing the final characteristics of the product in terms of structure, aroma, and digestibility. Leavening and maturation are therefore distinct but closely interconnected processes in determining the quality of the dough and the baked product.
16. Too much hydrolysis and loss of structure [4]
Proteolysis represents an important process in dough maturation, since the partial degradation of gluten proteins can improve the extensibility and workability of the dough. However, when protein hydrolysis becomes excessive, it can compromise the stability of the gluten network.
If proteolysis is too intense, the protein chains of glutenins and gliadins are progressively fragmented, reducing the number of intermolecular interactions that maintain the stability of the gluten network structure. Consequently, the protein network progressively loses elasticity and cohesion capacity.
When this process becomes marked:
• the gluten network collapses or weakens significantly
• the dough loses the ability to retain the gases produced during fermentation
• the structure of the dough becomes softer and less stable.
This phenomenon can occur, for example, under conditions of very long fermentations, high proteolytic activity, or strong acidification of the dough. In these cases, excessive degradation of proteins can lead to a less resistant structure, with negative effects on the volume and consistency of the final product.
17. Role of flour strength (W) [1].
Flour strength, indicated by the parameter W, is a measure of the flour’s ability to develop and maintain an elastic and resistant gluten network during mixing and fermentation. This value is determined using the Chopin alveograph and reflects the dough’s ability to retain the gases produced during fermentation. In general, flours with a high W value contain a greater amount of gluten proteins (gliadins and glutenins) and are able to form a more structured and resistant gluten network.
Strong flours are characterized by:
• higher protein content
• greater water absorption capacity
• greater resistance to dough deformation
These flours are often used for long-fermentation doughs, since their protein structure allows stability and gas retention capacity to be maintained over time. Conversely, flours with lower W form a less resistant gluten network and are generally more suitable for doughs with shorter fermentation times or for baked products that require a more crumbly structure.
18, How much gluten is hydrolyzed [2][5]
The degradation of gluten proteins during dough fermentation is generally limited in traditional baking processes.
Experimental studies indicate that:
• fermentation with baker’s yeast
4–6 hours → about 2–5% hydrolysis of gluten proteins
• longer fermentations (12–24 hours) → about 5–10% protein degradation
In sourdough fermentation systems, the combined presence of lactic acid bacteria and microbial proteolytic systems can increase the hydrolysis of gluten proteins, in some cases leading to higher values of protein degradation, especially during prolonged fermentations and controlled experimental conditions. However, in most baked products obtained with traditional methods, gluten degradation remains partial and the protein structure continues to perform its technological role in dough formation.
19. Digestion of gluten [10]
Gluten digestion is a complex process that involves several phases of the gastrointestinal tract and the combined action of digestive enzymes and the microbiota.
The process can be divided into four main phases:
-
Oral cavity
During chewing, gluten is mechanically fragmented and mixed with saliva. Although saliva mainly contains amylolytic enzymes, the oral microbiota may contribute to a limited extent to the initial degradation of some peptides.
-
Stomach
In the stomach, gluten proteins are partially hydrolyzed by the enzyme pepsin, active in an acidic environment, generating smaller polypeptides.
-
Small intestine
In the duodenum and jejunum, pancreatic enzymes intervene, particularly trypsin, chymotrypsin and elastase, which continue protein digestion producing smaller peptides. However, due to the high content of proline and glutamine, some gluten sequences remain resistant to complete digestion.
-
Intestinal microbiota
Peptides not completely digested can be further metabolized by the intestinal microbiota, which possesses proteolytic enzymes capable of degrading some gluten fragments.
The The microbiota contributes to peptide degradation [10].
20. Actual digestibility and perception [4]
The perception of “lightness” of a baked product does not depend exclusively on the biochemical digestion of its components, but also on structural and compositional factors of the product.
Among the elements that can influence this perception are:
• alveolation
• dough hydration
• fat content
Good alveolation makes the structure of the product more porous and less compact, facilitating fragmentation during chewing and the action of digestive enzymes. Dough hydration can influence the perception of digestibility because more hydrated doughs tend to develop a more open and less dense structure after baking. This softer and more alveolated structure generally requires less mechanical work during gastric digestion, and is therefore perceived as lighter. In addition, greater hydration favors more intense enzymatic processes during fermentation (on starches and proteins), which can contribute to a partial degradation of macromolecules before baking. Finally, the fat content of the meal can influence digestion by slowing gastric emptying, increasing the sensation of heaviness or fullness after consumption. Overall, the perception of digestibility of a baked product is therefore the result of the interaction between food structure, nutritional composition, and the individual response of the digestive system.
21. The 72-hour myth [6]
In the context of baking, the idea is often widespread that very long fermentation times, such as 48–72 hours, are always necessary to obtain a more digestible or better matured dough. In reality, numerous studies show that much of the biochemical transformation of dough occurs in the first 18–24 hours of fermentation [6].
During this initial phase, the main processes of enzymatic maturation occur, including:
• the activity of amylases, which degrade starch producing fermentable sugars;
• the action of proteases, which partially hydrolyze gluten proteins;
• the metabolic activity of yeasts and lactic acid bacteria, which produce organic acids, CO₂ and aromatic compounds.
These processes contribute to modifying the structure of the dough, improving its workability, development of the gluten network and sensory characteristics of the final product.Extending fermentation beyond 24–36 hours may continue to influence some transformations, but with progressively more limited effects, while the risks of excessive acidification or weakening of the gluten network increase. For this reason, very long times such as 72 hours are not always necessary, and their effectiveness depends on several factors, including type of flour, fermentation temperature, hydration and microbiota present in the dough.
22. Why some pizzas cause bloating [9][11]
The sensation of bloating after the consumption of pizza may depend on several factors related both to the composition of the dough and to the way the meal is consumed.
One One possible cause is the presence of FODMAPs, particularly fructans derived from wheat. These carbohydrates may be poorly digested in the small intestine and subsequently fermented by the intestinal microbiota, with gas production that can cause bloating especially in sensitive individuals.
Another important factor is the quantity consumed and the overall composition of the meal. Very abundant meals or meals rich in fats can slow gastric emptying and make digestion slower, favoring the sensation of heaviness or abdominal distension.
The characteristics of the flour used can also influence the digestibility of pizza. The use of very strong flours, rich in proteins and with high baking strength, leads to the formation of a more tenacious gluten network, which may be slower to digest if the dough does not undergo adequate enzymatic maturation.
In addition, the times and conditions of fermentation and maturation of the dough play a relevant role. Fermentation that is too short may not allow sufficient enzymatic activity (amylases and proteases), with less degradation of starches and proteins. Conversely, adequate fermentation processes favor the partial hydrolysis of macromolecules, improving the workability of the dough and the tolerability of the final product.
Finally, individual factors, such as sensitivity to FODMAPs, the composition of the intestinal microbiota and digestive functionality, can also influence the appearance of bloating after the consumption of pizza.
A further element to consider is dough hydration. Doughs with low hydration tend to be more compact and less digestible, whereas greater hydration favors enzymatic activity, a more alveolated structure and generally faster digestion. Very dry doughs may instead be denser and heavier in the stomach, contributing to the sensation of bloating.
Pizza baking also affects digestibility. Adequate baking allows starch gelatinization and protein denaturation, making the structure of the food more easily digestible. Conversely, an undercooked pizza may contain partially gelatinized starch and therefore be less digestible, favoring phenomena of intestinal fermentation and gas production.
Overall, bloating associated with pizza consumption does not depend on a single factor, but on the interaction of several technological and nutritional elements, including type of flour, dough hydration, maturation and fermentation times, baking method, quantity consumed and individual characteristics of the consumer.
23. Role of the intestinal microbiota [10]
The intestinal microbiota consists of a complex community of microorganisms that plays an important role in the digestive and metabolic processes of the organism. In addition to the enzymes produced by the human digestive system, many intestinal bacteria possess specific enzymes capable of degrading food compounds that are not completely digested. In particular, the microbiota contributes to the fermentation of non-digestible carbohydrates and to the degradation of some residual peptides and proteins, producing metabolites such as short-chain fatty acids (SCFAs), including acetate, propionate and butyrate. These compounds perform several physiological functions, including supporting the metabolism of intestinal cells and modulating some immune processes. The composition of the microbiota can therefore influence individual tolerance to different foods, including wheat-based products, contributing to variability in the digestive response among different individuals [10].
24. Gluten or fructans [11]
For a long time, gastrointestinal symptoms associated with the consumption of wheat-based products were mainly attributed to gluten. However, more recent clinical studies suggest that, in many cases, such symptoms may be caused by other wheat components, particularly fructans. Fructans belong to the category of FODMAPs, fermentable carbohydrates that may be poorly absorbed in the small intestine and subsequently fermented by the intestinal microbiota in the colon. This fermentation process can lead to the production of gas and organic acids, causing symptoms such as bloating, abdominal pain and distension. Some controlled clinical studies have shown that, in subjects with non-celiac wheat sensitivity, gastrointestinal symptoms are often more associated with fructans than with gluten itself [11].
25. Reduction of FODMAPs with sourdough [9]
Sourdough fermentation can contribute to the reduction of FODMAP content, particularly of the fructans present in wheat flour. During lactic fermentation, some lactic acid bacteria possess enzymes capable of degrading fructans into simpler sugars, which are subsequently metabolized by the microorganisms present in the dough. This process leads to the production of lactic acid, acetic acid and other fermentative metabolites. In sufficiently long fermentations, this microbial metabolism can lead to a significant reduction in fructan content, potentially contributing to improved tolerability of baked products for some people sensitive to FODMAPs [9]. The effectiveness of this reduction, however, depends on several factors, including fermentation time, composition of the sourdough microbiota, type of flour and fermentation conditions.
26. Individual variability [10] [11]
The digestion and tolerability of wheat-based products can vary among individuals and depend on several factors related both to the food and to digestive conditions.
Among the most relevant factors are:
• quantity of food consumed
• structure of the product (alveolation, hydration, degree of baking)
• composition of the meal, particularly the presence of fats or other foods that may influence gastric emptying.
The characteristics of the dough and the fermentation process, such as the type of flour used, the level of hydration and the maturation times, can also influence the final structure of the product and therefore the way it is digested. For this reason, the perception of digestibility of baked products can vary considerably depending both on the technological characteristics of the product and on the conditions of consumption.
Specific studies
1 – Skodje et al., 2018
Fructan, rather than gluten, induces symptoms in patients with self-reported non-celiac gluten sensitivity. This randomized double-blind clinical study compared the effects of gluten, fructans and placebo in subjects with self-reported non-celiac gluten sensitivity. The results showed that fructans induced significantly greater gastrointestinal symptoms compared with gluten. This suggests that, in many cases, symptoms attributed to gluten may be related to the fermentation of FODMAPs by the intestinal microbiota. The study therefore highlights the role of individual variability in digestive response and the interaction between diet and microbiota in the onset of intestinal symptoms.
2. Flint et al., 2012
The role of the gut microbiota in nutrition and health
The scientific review describes the role of the intestinal microbiota in the digestion of carbohydrates and proteins not completely digested by the human body. Intestinal bacteria possess numerous metabolic enzymes that allow the fermentation of residual food substrates, producing metabolites such as short-chain fatty acids (SCFAs). Differences in microbiota composition among individuals can therefore influence digestion, nutrient absorption and tolerance to specific foods.
27. Diagram of biochemical processes [4][5]
Simplified sequence:
Flour + water
↓
protein hydration
↓
gluten formation
↓
amylases → sugars
↓
yeast → CO₂
↓
proteases → gluten degradation.
28. Main chemical reactions
Alcoholic fermentation
glucose → CO₂ + ethanol.
Lactic fermentation
glucose → lactic acid.
29. Evolution of dough structure [4][5]
The structure of the dough is not static, but evolves progressively during the different phases of processing and fermentation. This evolution depends on the interaction between physical, enzymatic and microbiological processes, which modify over time the organization of the gluten network and the distribution of gases in the dough.
The main phases of this evolution can be schematized as follows.
1. Gluten formation
During mixing, the hydration of wheat proteins (gliadins and glutenins) allows the formation of the gluten network, an elastic three-dimensional structure that gives the dough cohesion, elasticity and extensibility. This network represents the framework that will allow the gases produced during fermentation to be retained.
2. Fermentation
During fermentation, yeasts metabolize sugars producing carbon dioxide (CO₂) and ethanol. The gas produced accumulates within the gluten network forming gas bubbles that determine dough expansion and the development of alveolation.
3. Enzymatic maturation
Alongside fermentation, enzymatic maturation processes occur. The enzymes naturally present in flour, particularly amylases and proteases, progressively degrade starches and proteins. Amylases produce fermentable sugars, while proteases partially modify the gluten network, making the dough more extensible and workable.
4. Possible protein degradation
If fermentation times are further prolonged, protease activity can become more marked, leading to more extensive degradation of gluten proteins. In this phase some peptide bonds are progressively hydrolyzed, with consequent weakening of the gluten network.
The phenomenon can have different effects depending on the intensity of the process. Moderate degradation can improve dough workability and contribute to maturation. Conversely, excessive proteolysis can compromise the ability of the gluten network to retain gases, leading to a weaker and less stable dough.
Overall, the evolution of dough structure is the result of a dynamic balance between formation, strengthening and progressive modification of the protein network, influenced by factors such as fermentation time, temperature, hydration and flour strength.
30. Experimental percentages of gluten degradation [5][8]
Several experimental studies have evaluated the extent of gluten protein degradation during dough fermentation.
The results generally show that:
• short fermentation with baker’s yeast
≈ 2–5% protein hydrolysis
• longer fermentations (12–24 hours)
≈ 5–10% gluten degradation
• fermentations with sourdough and selected lactic acid bacteria
up to 20–40% protein degradation under controlled experimental conditions.
It is important to emphasize that these values depend on numerous factors, including:
• fermentation time
• composition of the sourdough microbiota
• temperature and pH of the dough
• activity of microbial proteolytic systems.
Under standard baking conditions, gluten degradation therefore remains partial and rarely leads to complete hydrolysis of the proteins.
31. Conclusions [4][5][7][9][10]
The digestibility of doughs and baked products is the result of a complex series of biochemical, microbiological and technological processes that occur during dough preparation, fermentation and baking. It therefore does not depend on a single factor, but on the interaction between different elements that influence the structure and transformation of the macromolecules present in flour.
Among the most relevant factors are:
• the time and conditions of fermentation, which determine the intensity of dough maturation processes
• the natural enzymatic activity of flour, particularly that of amylases and proteases, which contributes to the partial degradation of starches and proteins
• the presence of lactic acid bacteria, which can favor further biochemical transformations, including dough acidification, proteolytic activity and reduction of some components such as fructans
• the structural characteristics of the dough, influenced by technological factors such as hydration, flour strength and fermentation method
• the individual variability of digestion, which may also be influenced by the composition of the intestinal microbiota.
Overall, enzymatic maturation processes play a central role in modifying dough structure before baking, contributing to the transformation of macromolecules and the formation of the final characteristics of the product.
It is also important to emphasize that baker’s yeast (Saccharomyces cerevisiae) mainly has the role of producing carbon dioxide (CO₂) responsible for leavening, while its direct contribution to the degradation of gluten proteins is relatively limited compared with that of flour enzymes and the proteolytic systems present in lactic acid bacteria.
Therefore, the digestibility of baked products cannot be attributed exclusively to the type of yeast used, but must be interpreted as the result of the balance between fermentation, enzymatic maturation, dough structure and process conditions that characterize dough production.
Bibliografia
[1] Shewry, P. R., Halford, N. G., Belton, P. S., & Tatham, A. S. (2002).
The structure and properties of gluten: an elastic protein from wheat grain.
Philosophical Transactions of the Royal Society B, 357, 133–142.
[2] Wieser, H. (2007).
Chemistry of gluten proteins.
Food Microbiology, 24, 115–119.
[3] Don, C., Lichtendonk, W. J., Plijter, J. J., & Hamer, R. J. (2003).
Glutenin macropolymer: a gel formed by glutenin particles.
Journal of Cereal Science, 37, 1–7.
[4] Belitz, H. D., Grosch, W., & Schieberle, P. (2009).
Food Chemistry. Springer, Berlin.
[5] Coda, Di Cagno, Gobbetti e Rizzello (2014).
Sourdough lactic acid bacteria: exploration of non-wheat cereal-based fermentation.
Food Microbiology, 37, 51–58.
[6] Corsetti, A., & Settanni, L. (2007).
Lactobacilli in sourdough fermentation.
Food Research International, 40, 539–558.
[7] De Vuyst, L., & Neysens, P. (2005).
The sourdough microflora: biodiversity and metabolic interactions.
Trends in Food Science & Technology, 16, 43–56.
[8] De Angelis et al. (2006) con titolo su gliadin polypeptides responsible for celiac sprue, pubblicata in Biochimica et Biophysica Acta
[9] Loponen, J., & Gänzle, M. (2018).
Use of sourdough in low FODMAP baking.
Foods nel 2018, 7(7):96
[10] Flint, H. J., Scott, K. P., Louis, P., & Duncan, S. H. (2012).
The role of the gut microbiota in nutrition and health.
Nature Reviews Gastroenterology & Hepatology, 9, 577–589.
[11] Skodje, G. I., Sarna, V. K., Minelle, I. H., et al. (2018).
Fructan, rather than gluten, induces symptoms in patients with self-reported non-celiac gluten sensitivity.
Gastroenterology, 154, 529–539.